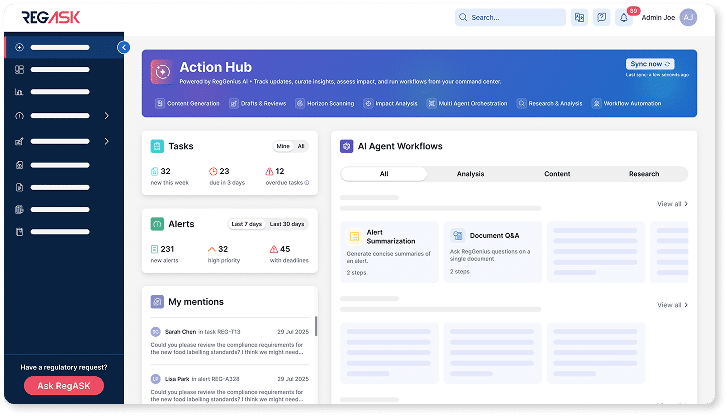
Command Center
Control regulatory operations from one place
Track change, manage workflows, and trigger actions from a single view

Turn regulatory change into coordinated, auditable action. RegASK elevates regulatory professionals with speed, traceability, and control.


RegASK is the AI system for modern regulatory work. Powered by Agentic AI, human expertise, and enterprise governance, it delivers triaged regulatory alerts, tailored impact assessments, and end-to-end workflow orchestration.
Teams can improve compliance outcomes, mitigate risk, and accelerate time-to-market across 160+ countries.
Horizon scanning across 160+ markets, AI-driven regulatory intelligence, impact assessments, and prioritized alerts. Your team always sees what’s coming before it arrives.
Workflow orchestration, structured assessments, labelling compliance, and prioritized actions. Turn regulatory intelligence into speed-to-market.
Open API, MCP support, pre-built connectors, and enterprise SSO for Veeva Vault and Esko make RegASK the regulatory intelligence layer across your entire ecosystem.
Regulatory monitoring, document management, full audit trail, and direct access to 1,800+ subject matter experts. Every compliance decision is traceable and defensible.
BUILT WITH YOUR TEAM IN MIND

Command Center
Control regulatory operations from one place
Track change, manage workflows, and trigger actions from a single view

Monitoring
See regulatory change as it happens
Near real-time tracking across 160+ markets, prioritized by product, market, and business relevance
Intelligence
Turn regulatory signals into structured insight
AI agents analyze, summarize, and classify content, accelerating the path from signal to decision
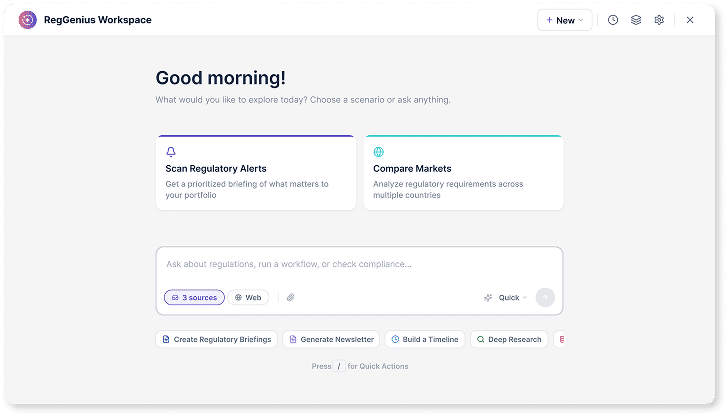
Regulatory Assistant
Ask regulatory questions and get cited answers instantly
Interact in natural language and receive contextual, source-grounded responses

Expert Validation
Validate critical insights with human expertise
1,800 Regulatory experts-in-the-loop add local context, review, and confidence where it matters most

Workflows
Move from insight to coordinated action
Assess impact, assign tasks, and manage execution through structured workflows and timelines
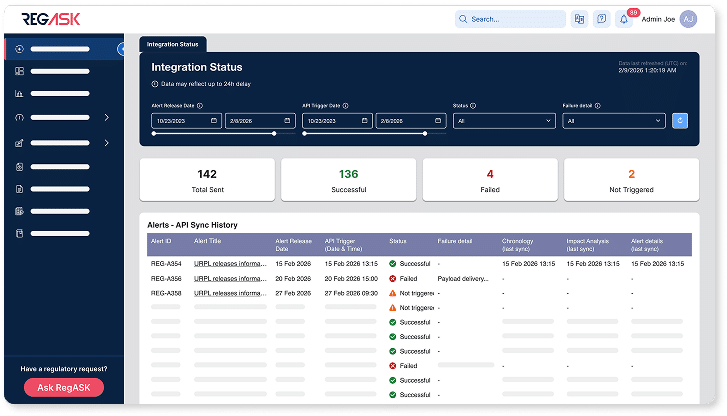
Integrations
Connect regulatory intelligence to your systems
Deliver structured outputs into Veeva Vault RIM, QMS, labeling, and product lifecycle workflows
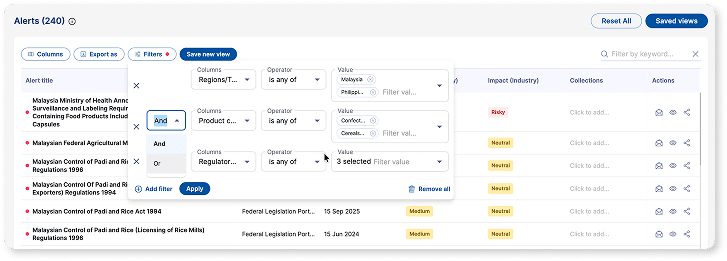
Content Curation
Cut through noise and focus on what matters
Curated, prioritized content tailored to your products, markets, and risk areas
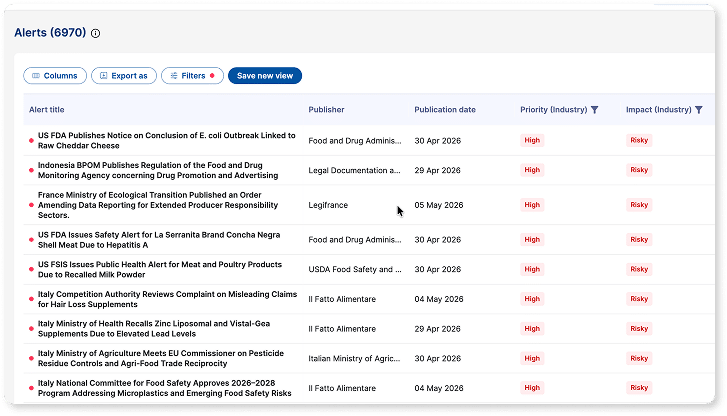
Risk Management
Identify and manage regulatory risk early
Detect risks before they impact compliance, timelines, or product launches

Impact Assessment
Understand how regulatory change affects your business
Assess impact across products, markets, and functions in a single view
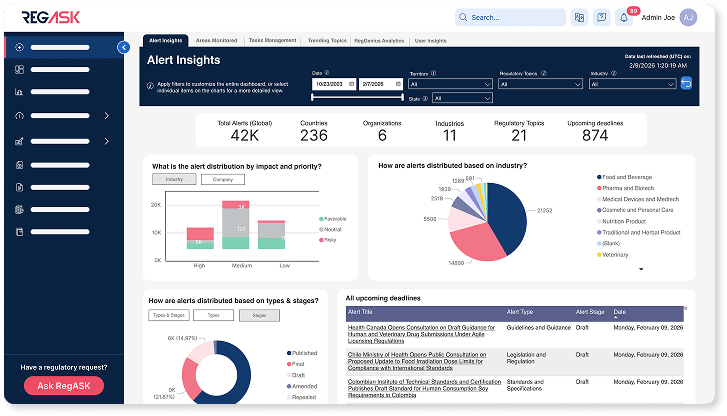
Insights
Turn regulatory data into decision-ready insight
Spot trends, surface risks, and inform leadership with clarity
Our infrastructure is designed to provide a secure, compliant, and resilient service environment, helping your teams meet regulatory compliance needs with precision and reliability.







Hosted on Microsoft Azure, RegASK benefits from one of the most secure and reliable cloud platforms in the industry. Azure’s comprehensive compliance coverage helps address regulatory requirements, and its built-in security protects applications and data from external threats.
Our security infrastructure is designed to protect data against unauthorized access or modification while ensuring it remains accessible and available. We employ state-of-the-art encryption and secure data handling practices, leveraging tools like Microsoft Defender to provide ongoing surveillance of our systems to detect and prevent potential security threats.
Experience AI-driven regulatory intelligence and workflow orchestration